Access to Drugs Before FDA Approval: Video Explainer with Christopher Robertson
In this video explainer, Christopher Robertson discusses the Right to Try Act and off-label use of pharmaceuticals with Alison Bateman-House.

In this video explainer, Christopher Robertson discusses the Right to Try Act and off-label use of pharmaceuticals with Alison Bateman-House.

The question must be: can we accommodate non-emergent/non-urgent care safely or not, and if yes, which care do we address first as we re-open?

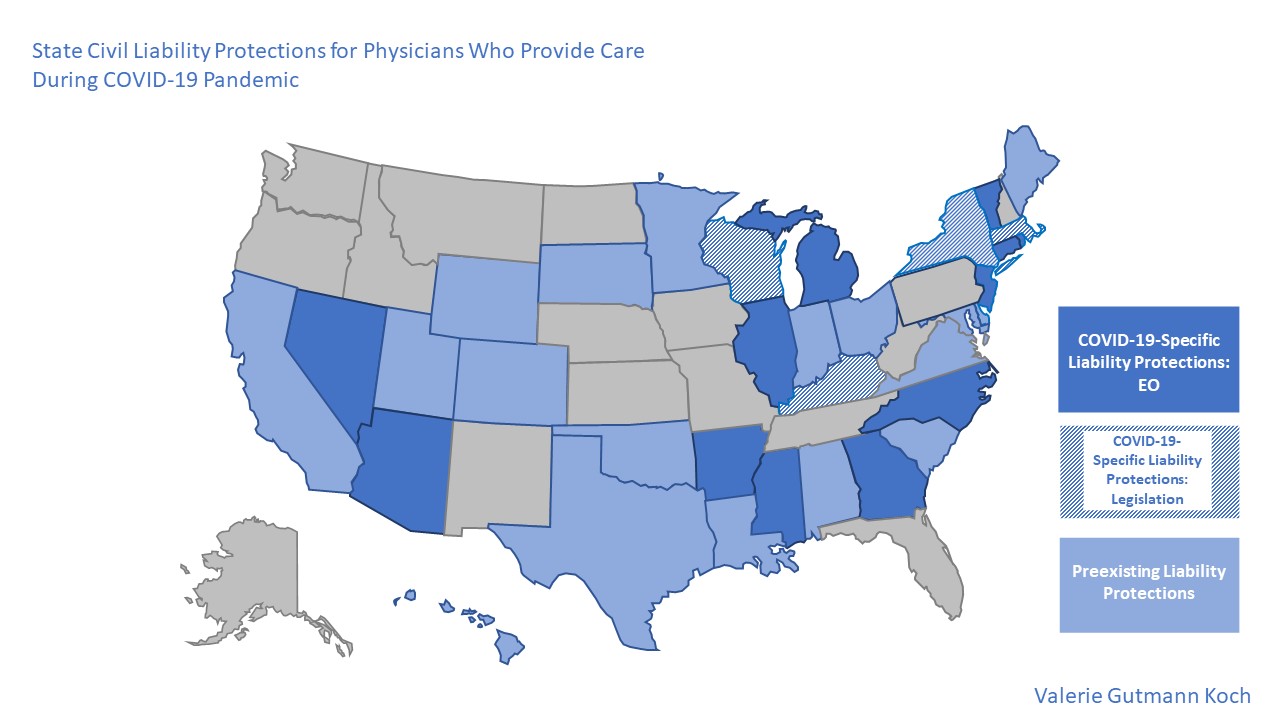
Clinicians and policymakers alike are raising the alarm about potential legal liability for following crisis standards of care during the COVID-19 pandemic.
This was the first time in a long time that I’ve renewed my nursing license with the thought that I might need it — that I might be needed.

In addition to tracing the early history of the Missouri and New York suits, we explain how these lawsuits are being used as conduits for misinformation.

Until there is a vaccine or treatment for COVID-19, our economy and privacy will be at the mercy of imperfect technology used in the pandemic response.

Government authorities must be extremely cautious about basing public policy decisions on inadequately vetted findings, no matter how much hype they get.
Almost immediately, there was a public outcry about the possibility that this was an illegal or unethical “research experiment.”
