Year in Review: Biosimilars
In November 2020, the FDA released its most recent draft guidance for industry on biosimilarity and interchangeability
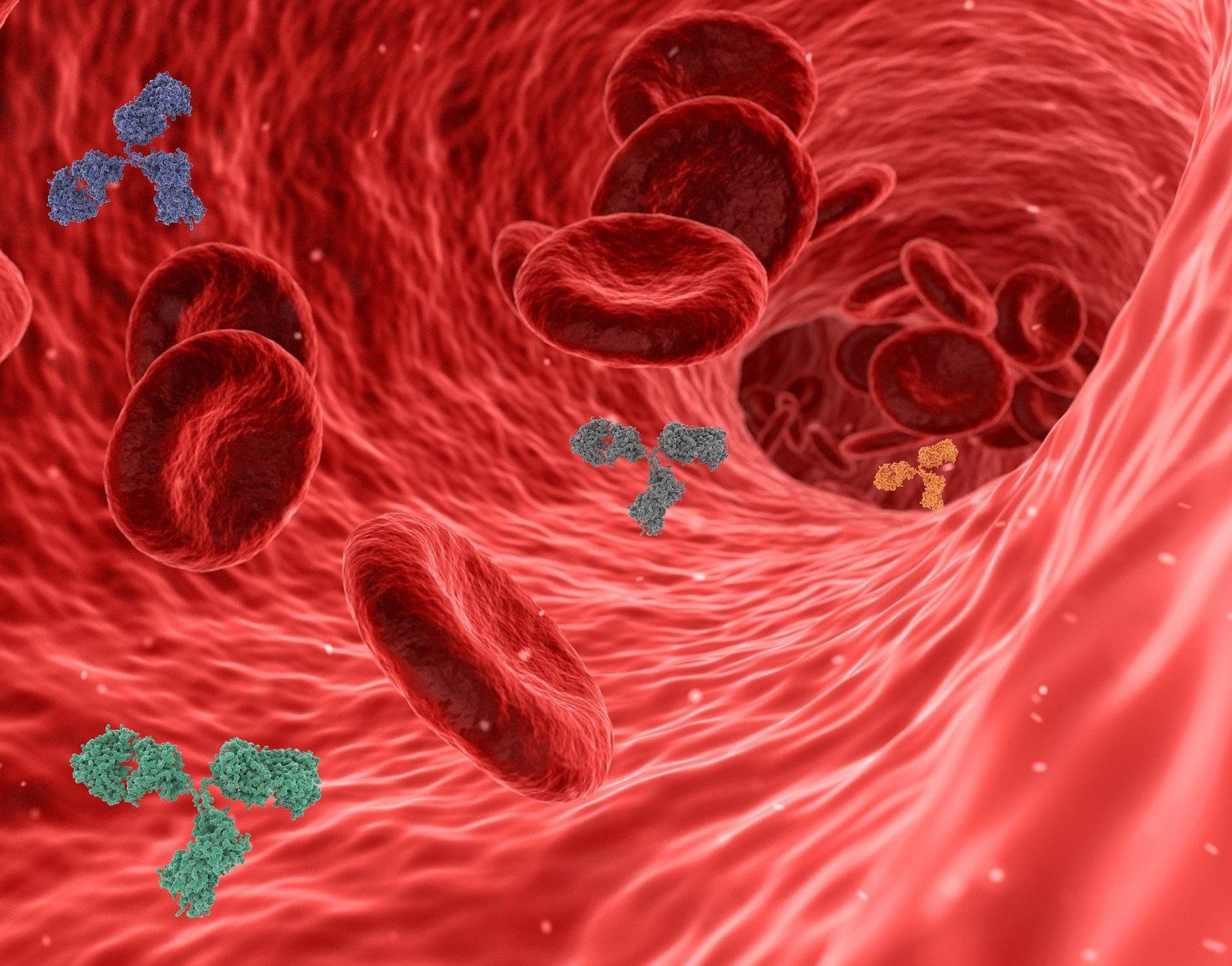
In November 2020, the FDA released its most recent draft guidance for industry on biosimilarity and interchangeability

The purpose of the secondary patent filings was to assemble a thicket of patents, 132 in all, to prohibit competition from biosimilar companies.

Procedurally, the Trump administration’s issuance of the Most Favored Nation Interim Final Rule was a mistake.

Selections feature topics ranging from political pressures facing the FDA, to the financial incentive structure for antibiotic development.
Interesting empirical studies, policy analyses, and editorials on pharma, health law, and policy issues from August 2020.

Interesting empirical studies, policy analyses, and editorials on health law and policy issues from July 2020.
While generic competition is crucial for reducing drug prices, brand-name drug manufacturers can utilize several strategies to delay such competition.

Topics include the association between the clinical benefit of cancer drugs and their prices, and whether to include children in COVID-19 therapy trials.

By Rahul Nayak Interested in learning more about pharmaceutical policy? Curious about the role of the FDA in ensure safe and effective drugs reach the market? Wondering why drug prices are so high in the US? Readers of my prior posts may enjoy learning more about these topics! Check out a free HarvardX online course,…

While many issues are likely to play a prominent roles in this campaign, healthcare is likely to play an outsized role.
