Can Schools Require the COVID-19 Vaccine? Education, Equity, and the Courts
Given the potential legal challenges policymakers may face, legislation to remedy the COVID-19 educational crisis must be carefully drafted.

We explain why COVID-19 vaccines are only just starting to be tested in children and what policymakers can do to spur pediatric vaccine trials.
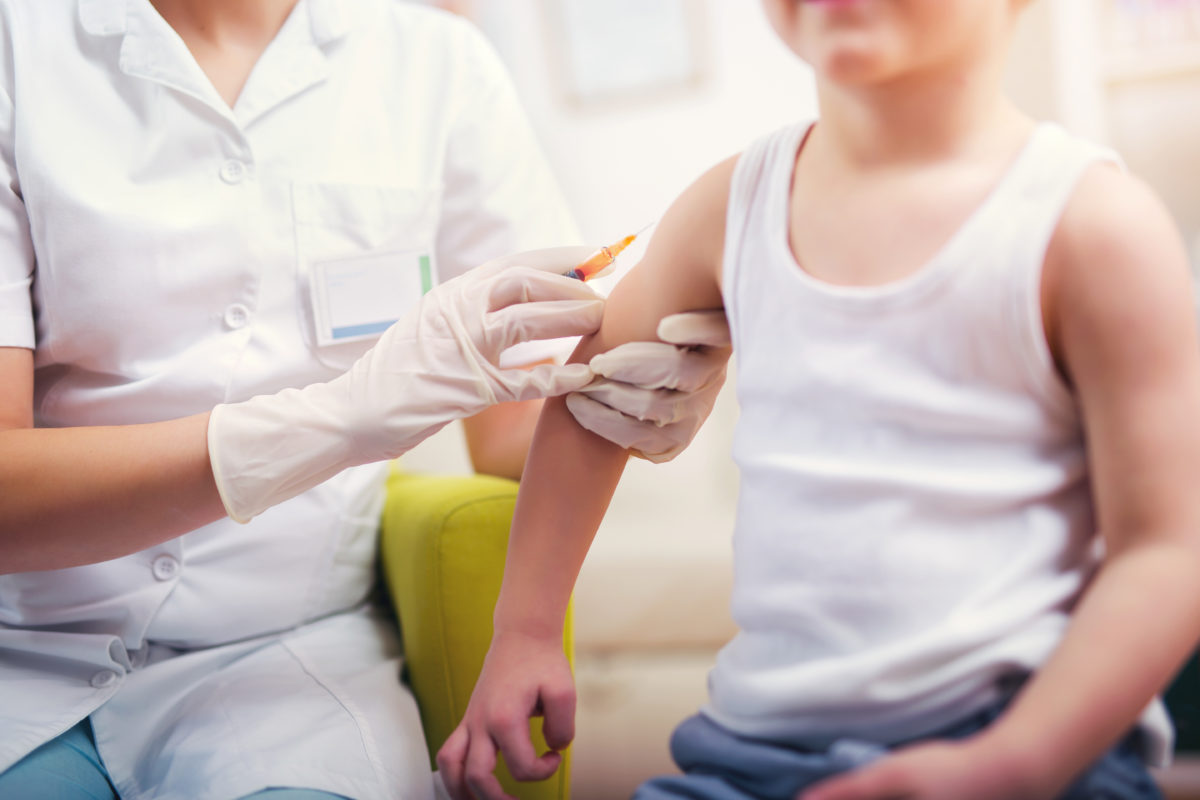
The study, which involved an online survey of and interviews with older adults, revealed a heterogeneity of ways of thinking about capacity.

This blog post provides an overview of the FDA’s emergency authorization powers and analyzes the extent of their usage in the COVID-19 pandemic.

Pharmaceutical companies are now also engaging the public themselves in an attempt to build trust in their products.
Selections feature topics ranging from political pressures facing the FDA, to the financial incentive structure for antibiotic development.
The lack of diversity among research participants can have significant consequences on generalizability and distribution of treatments.

The field has a frustrating tendency to make lofty claims about psychedelics that stray from the realities and limitations of the data.
Interesting empirical studies, policy analyses, and editorials on pharma, health law, and policy issues from August 2020.
